In the News
All in the Family: What Multigenerational Cohorts Are Revealing about Potential Environmental Impacts on Neurodevelopment
-
Focus Areas
Environmental Health

Abstract
Right after giving birth to her daughter in 1962, Marcy Hartman—a newspaper reporter at the time—watched a nurse remove a syringe full of blood from the placenta. “It was a small contribution I made to the future,” remembers Hartman, now 75.
Between 1959 and 1967, Hartman and more than 15,000 other pregnant women in the San Francisco Bay Area enrolled in a long-term cohort called the Child Health and Development Studies (CHDS).1 The cohort was intended to help scientists better understand what makes pregnancies, and children, healthy. Researchers are now using the biological specimens and other data provided decades ago by Hartman and the other cohort members to tackle one of the most vexing scientific questions about the role of the environment in health—whether an individual’s exposures can impact the health of his or her descendants.
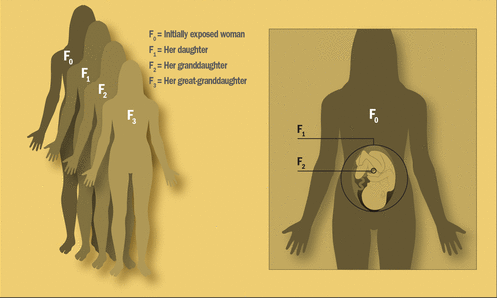
Unlike males, who make sperm throughout life, females are thought to be born with all the eggs they will ever produce. That means that each prenatal exposure has the potential to directly impact three generations, says epidemiologist Barbara Cohn, principal investigator of the CHDS. They include the mother (known as the F0 generation), the fetus (F1 generation), and, if the fetus is a girl, all her immature egg cells—any of which may one day become the F2 generation.
When a pregnant woman is exposed to an environmental agent, the exposure can affect not only herself () and her unborn child (F1) but also the germ cells developing within the fetus, if that fetus is a girl (F2). The woman’s great-grandchild (F3) is the first generation not directly affected by the original exposure. Image: EHP.
In the past decade, studies in laboratory animals have pointed to the possibility that exposures to potentially toxic environmental chemicals and other stressors in utero may affect normal development of not just the growing fetus, but multiple generations to come.2 In one mouse study, for instance, the descendants of mice treated during pregnancy with di-(2-ethylhexyl) phthalate had lower baseline and stress-induced plasma levels of the stress hormone corticosterone than untreated mice.3 Another study showed differences in the social interactions of mice of the and generations of dams fed bisphenol A compared with those of unexposed dams.4
Unsurprisingly, evidence from human studies has proved elusive.5 People, after all, reproduce far more slowly than mice, and keeping tabs on thousands of humans for decades is no small feat. There are very few cohorts in the world that are able to generate the kind of long-term, multigenerational data that would be needed to address questions about the potential cross-generational effects of human exposures. Yet the few that do exist are beginning to yield clues about how a person’s exposures and experiences could influence the neurodevelopment and behavior of future generations.6,7
Findings in Humans Emerge
In 2017, researchers studying a large group of parents and children in the United Kingdom reported associations between smoking by grandmothers during pregnancy and autism symptoms and diagnoses in grandchildren.6 Study leader Jean Golding, an epidemiologist at the University of Bristol, had founded the Avon Longitudinal Study of Parents and Children (ALSPAC) in 1991 to answer questions about child development.8
Golding did not design the 14,000-family cohort around autism risk, specifically. “At the time, the incidence of autism was thought to be lower than 1 in 1,000 people,” remembers Golding. She did not think that 14,000 children would be a large enough number to address questions about such a rare disorder. In fact, she expected fewer than 10 cases of autism to be counted among the cohort children.
But Golding became interested in risk factors for autism after studies documented a fivefold increase over previous years in the incidence of autism in kids born in the United Kingdom between 1988 and 1995.9 Over the years, the ALSPAC researchers identified more than 200 study children with possible or diagnosed autism.
Golding and colleagues had reason to think there could be a link between children’s neurodevelopment and their grandparents’ smoking habits. Previously they had found preliminary evidence suggesting that among boys whose mothers smoked during pregnancy, average head circumference was smaller for boys whose fathers, too, were exposed prenatally, compared with boys whose fathers were not exposed prenatally. (No such evidence was found for girls, however, and average head circumferences of boys or girls exposed to maternal smoking in utero did not differ depending on whether their mothers were also exposed prenatally.)10
Grandparents were not included in the ALSPAC cohort, so researchers asked the study parents whether their own mothers had smoked during pregnancy. Doctors, for the most part, did not discourage smoking during pregnancy in the 1950s and 1960s, when many of the parents were born.
The researchers found that after adjusting for known autism risk factors, including mother’s education and socioeconomic status, children whose maternal grandmothers smoked were more likely to be diagnosed with autism than those whose maternal grandmothers did not smoke.6 However, this association was only apparent if the child was also exposed to maternal smoking in utero.
Then in 2018, an analysis of data from the Nurses’ Health Study II (NHS-II), which had followed a large group of U.S. women since the late 1980s, also found evidence of a multigenerational exposure–outcome association. Researchers reported that children whose grandmothers used diethylstilbestrol (DES) during pregnancy were more likely to be diagnosed with attention-deficit/hyperactivity disorder (ADHD).7 Specifically, 7.7% of mothers who reported being exposed to DES in utero also reported ADHD in their children versus 5.2% of mothers who did not report in utero DES exposure.
Read the rest of the article in Environmental Health Perspectives.
Originally published by Environmental Health Perspectives
More Updates
Work With Us
You change the world. We do the rest. Explore fiscal sponsorship at PHI.
Support Us
Together, we can accelerate our response to public health’s most critical issues.
Find Employment
Begin your career at the Public Health Institute.